400-998-5282
专注多肽 服务科研
400-998-5282
专注多肽 服务科研
His-Leu,血管紧张素 I 的 C 端二肽在血管紧张素转换酶 (ACE) 的作用下分裂,产生血管紧张素 II。另见 ACE 基材 Z-FHL 和 HHL。由 Cu(II)、His-Leu 和乙二胺(或 His)产生的复合物水解裂解 DNA 的磷酸二酯骨架。
编号:179515
CAS号:7763-65-7
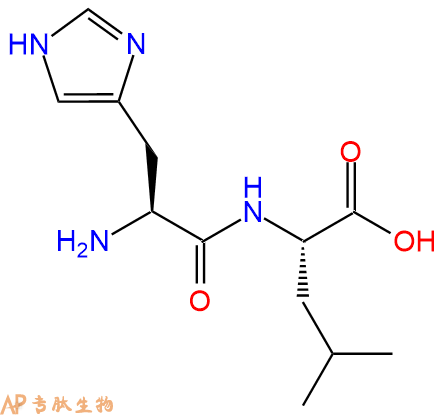
单字母:H2N-HL-OH
| 编号: | 179515 |
| 中文名称: | 二肽His-Leu |
| 英文名: | His-Leu |
| CAS号: | 7763-65-7 |
| 单字母: | H2N-HL-OH |
| 三字母: | H2N N端氨基:N-terminal amino group。在肽或多肽链中含有游离a-氨基的氨基酸一端。在表示氨基酸序列时,通常将N端放在肽链的左边。 -HisL-组氨酸:histidine。系统命名为(2S)-氨基-3-(4-咪唑基)丙酸。其侧链带有弱碱性的咪唑基,为编码氨基酸。是幼小哺乳动物的必需氨基酸。符号:H,His。 -LeuL-亮氨酸:leucine。系统命名为(2S)-氨基-4-甲基戊酸。是编码氨基酸。是哺乳动物的必需氨基酸。符号:L,Leu。 -OHC端羧基:C-terminal carboxyl group。在肽或多肽链中含有游离羧基的氨基酸一端。在表示氨基酸序列时,通常将C端放在肽链的右边。 |
| 氨基酸个数: | 2 |
| 分子式: | C12H20N4O3 |
| 平均分子量: | 268.31 |
| 精确分子量: | 268.15 |
| 等电点(PI): | - |
| pH=7.0时的净电荷数: | 1.21 |
| 平均亲水性: | -1.15 |
| 疏水性值: | 0.3 |
| 外观与性状: | 白色粉末状固体 |
| 消光系数: | - |
| 来源: | 人工化学合成,仅限科学研究使用,不得用于人体。 |
| 纯度: | 95%、98% |
| 盐体系: | 可选TFA、HAc、HCl或其它 |
| 生成周期: | 2-3周 |
| 储存条件: | 负80℃至负20℃ |
| 标签: | 600+种二肽(Dipeptide)现货 |
His-Leu, C-terminal dipeptide of angiotensin I is split off by action of angiotensin-converting enzyme (ACE) yielding angiotensin II. See also ACE substrates Z-FHL and HHL. The complex generated from Cu(II), His-Leu and ethylene diamine (or His) hydrolytically cleaves the phosphodiester backbone of DNA.
His-Leu is a signal peptide that contains the amino acid sequence His-Leu. It is used in a variety of diagnostic tests and has been shown to bind to the p-hydroxybenzoic acid inhibitor binding site on the enzyme histone H1. This peptide has also been shown to inhibit the activity of histone H1 enzymes, which are involved in the regulation of DNA replication and transcription. In addition, His-Leu has been shown to inhibit specific enzymes that regulate human serum albumin synthesis, such as preparative high performance liquid chromatography (Hplc) and terminal residues. His-Leu can be used for diagnosis or as an inhibitor for infectious diseases.
H-His-Leu-OH 是一种二肽,平衡极性与疏水表面,支持氢键与侧链堆积研究。咪唑基团提供金属结合与质子化多功能性。其紧凑结构允许短肽相互作用建模。应用包括生物物理分析、肽化学与结合研究。
H-His-Leu-OH is a dipeptide balancing polar and hydrophobic surfaces that supports studies of hydrogen bonding and side-chain packing. The imidazole group provides metal-binding and protonation versatility. Its compact structure enables modeling of short peptide interactions. Applications include biophysical analysis, peptide chemistry, and binding studies.
| DOI | 名称 | |
|---|---|---|
| 10.1080/09687860500093248 | Substrate preference is altered by mutations in the fifth transmembrane domain of Ptr2p, the di/tri-peptide transporter of Saccharomyces cerevisiae | 下载 |
| 10.1016/j.chembiol.2008.05.019 | Tryptophan aminopeptidase activity of several indole prenyltransferases from Aspergillus fumigatus | 下载 |
| 10.1590/s0100-879x2005000600007 | A continuous fluorescent assay for the determination of plasma and tissue angiotensin I-converting enzyme activity | 下载 |
| 10.1002/cbdv.200490066 | Copper(II) complexes containing N,N-donor ligands and dipeptides act as hydrolytic DNA-cleavage agents | 下载 |





