400-998-5282
专注多肽 服务科研
400-998-5282
专注多肽 服务科研
Melittin 是一种PLA2激活剂,可刺激低分子量 PLA2 的活性,而不会提高高分子量 PLA2 的活性。
编号:114268
CAS号:145854-61-1/20449-79-0/37231-28-0
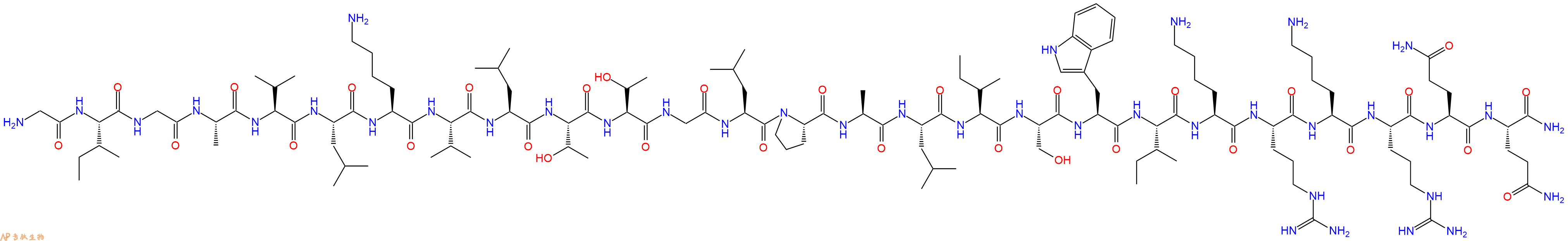
单字母:H2N-GIGAVLKVLTTGLPALISWIKRKRQQ-CONH2
| 参考文献(References): | Raynor et al., J. Biol. Chem., 266, 2753 (1991) Schweitz, Toxicon, 22, 308 (1984) |
Melittin TFA 是一种PLA2激活剂,可刺激低分子量 PLA2 的活性,而不会提高高分子量 PLA2 的活性。
Melittin TFA is a PLA2 activator, stimulates the activity of the low molecular weight PLA2, while it does not the increase activity of the high molecular weight PLA2[1][2].
在通常情况下,C-末端有4个氨基酸残基携带正电荷,N-末端有2个氨基酸残基携带正电荷,整个分子带6个正电荷。蜂毒肽的N-末端起前20个氨基酸残基主要是疏水的,C-末端的6个氨基酸残基主要是亲水的。分子的3个赖氨酸和2个精氨酸残基使其成为强碱性肽,在中性水溶液中,蜂毒肽作为单体是以随机的卷曲结构存在的,而随着pH值以及离子强度的增高,蜂毒肽自我交联,形成螺旋的四聚体结构,有研究发现在不同的溶液中蜂毒肽的螺旋结构区域及螺旋间的角度是不同的。螺旋结构中前21个氨基酸是极性的,位于螺旋的表面,而非极性氨基酸在螺旋的另一面。其两亲性(am-phiphilie)是膜结合肽和膜蛋白跨膜螺旋的特征。所以这个特性决定蜂毒肽既可以溶于水中,又可以与膜自然结合,进而溶解细胞。
Melittin蜂毒肽是由26个氨基酸残基组成的阳离子两亲抗菌多肽,它蜂毒中的主要功能物质,分子量为2846.46。蜂毒肽具有多种药理作用,比如蜂毒肽具有抗炎、镇痛作用,可以有效地治疗类风湿关节炎,对关节起保护作用。蜂毒肽也具有抗菌作用,在体外,蜂毒肽对MRSA菌株的作用比对其他革兰阳性菌更为显著,更利于增强MRSA诱导的伤口的愈合。蜂毒肽还具有抗病毒作用,其对艾滋病病毒具有杀伤作用等。此外它对抗肿瘤也有一定的效果。
蜂毒肽是从蜜蜂(Apis mellifera)的毒液中分离出来的一种多肽。它抑制蛋白激酶C、Ca2+/钙调蛋白依赖性蛋白激酶II、肌球蛋白轻链激酶和Na+/K+-ATP酶(突触体膜)。
Melittin is a polypeptide isolated from the venom of the honey bee (Apis mellifera). It inhibits protein kinase C, Ca2+/calmodulin-dependent protein kinase II, myosin light chain kinase and Na+/K+-ATPase (synaptosomal membrane).
蜂毒肽是蜂毒的主要成分,已被证明对革兰氏阳性细菌(包括耐甲氧西林金黄色葡萄球菌)具有抗菌功效。它还显示出抗病毒活性,例如它抑制人类免疫缺陷病毒和单纯疱疹病毒。此外,由于它抑制磷脂酶A2酶,因此可用于治疗炎症相关疾病。除此之外,蜂毒肽还具有抑制炎症介质如一氧化氮和环氧合酶-2的能力,有助于治疗炎症性疾病。由于蜂毒肽具有攻击脂质膜和抑制COX-2 mRNA表达的能力,因此它也可用于治疗肿瘤。蜂毒肽的结合形式对哺乳动物细胞无毒,但当它释放到细胞质中时是有毒的。蜂毒肽已被证明可抑制小鼠的转录调控和运动活性。这可能是由于其结合DNA的能力,阻止了某些基因的转录和翻译。最后,它具有很强的溶血活性,并且可以通过胰腺β细胞的去极化来增加胰岛素分泌。蜂毒肽是一种多样的肽,可用于整个研究和治疗领域。
Melittin is a major component of bee venom that has been shown to have antibacterial efficacy against gram-positive bacteria, including methicillin-resistant Staphylococcus aureus. It also demonstrates anti-viral activity, for example it inhibits human immunodeficiency virus and herpes simplex virus. Furthermore it can be used in the treatment of inflammatory-related illnesses due to it inhibiting the phospholipase A2 enzyme. In addition to this Melittins abaility to inhibit inflammatory mediators such as nitric oxide and cyclooxygenase-2 aids in the treatment of inflammatory diseases. As a result of Melittin having the capabilities of attacking lipid membranes and it surpressing COX-2 mRNA expression it can also be used in the treatment of tumours. The bound form of melittin is not toxic to mammalian cells, but it is toxic when it is released into the cell cytoplasm. Melittin has been shown to inhibit transcriptional regulation and locomotor activity in mice. This may be due to its ability to bind DNA, preventing transcription and translation of certain genes. Lastly It has strong hemolytic activity and been seen to increase insulin secretion via depolarization of pancreatic beta-cells. Melittin is a diverse peptide which can be used ina whole spectrum of research and therapeutic areas.
蜂毒肽是一种26个残基的肽,最初是从欧洲蜜蜂的毒液中分离出来的。蜂毒肽是一种来自蜜蜂毒液的阳离子溶血肽。蜂毒肽降低质膜的表面张力并引起细胞裂解。蜂毒肽具有强大的抗炎和抗菌活性。蜂毒肽已被广泛用作观察膜脂蛋白相互作用的模型肽。
Melittin is a 26-residue peptide originally isolated from venom of the European honeybee. Melittin is a cationic, hemolytic peptide from honey bee venom. Melittin lowers the surface tension at the plasma membrane and causes cell lysis. Melittin exhibits potent anti-inflammatory and antimicrobial activity. Melittin has been extensively used as a model peptide for observing membrane lipid-protein interactions.
从蜂毒中分离出的26个残基多肽,以Ca2+依赖性方式结合钙调蛋白,激活磷脂酶A2,并通过以Mg-ATP敏感的方式结合催化结构域来抑制蛋白激酶C(IC50=5-7 m m)。IC50为1-4μM。
26-residue polypeptide isolated from bee venom that binds calmodulin in a Ca2+ dependent manner, activates phospholipase A2, and inhibits protein kinase C (IC50 = 5-7 m M) by binding to the catalytic domain in a Mg-ATP sensitive manner. IC50 of 1-4 µM.
Definition
Protein kinases are transferase that catalyze the phosphorylation of proteins by covalently attaching phosphate groups to them, using ATP as a phosphate donor. Reversible protein phosphorylation-dephosphorylation has a principal role in the regulation of essentially all cellular functions. Kinase/phosphatase substrates can be found grouped according to their kinase families. A single substrate can have a many number of modifications.
Discovery
Phoebus AL at the Rockefeller Institute identified phosphate in the protein Vitellin (phosvitin) in 1906 1 and by 1933 Fritz Lipmann had detected phosphoserine in Casein 2. In 1954 Eugene P. Kennedy described the first ‘enzymatic phosphorylation of proteins’ in a variety of normal and malignant tissues, he showed that the phosphorus of the phosphoprotein fraction undergoes a high rate of turnover, as measured by incorporation of 32P 3 .
Structural Characteristics
There are thousands of different kinds of proteins in any particular cell that are substrate for different kinases and phosphatases. Phosphorylation of any site on a given protein can change the structure, function or localization of that protein. Within a protein, phosphorylation can occur on several amino acids. Phosphorylation on serine is the most common, followed by threonine. Tyrosine phosphorylation is relatively rare. However, since tyrosine phosphorylated proteins are relatively easy to purify using antibodies, tyrosine phosphorylation sites are relatively well understood. Histidine and aspartate phosphorylation occurs in prokaryotes as part of two-component signaling and in some cases in eukaryotes in some signal transduction pathways 4. Phosphorylation of seryl or threonyl (and occasionally tyrosyl) residues triggers small conformational changes in these proteins that alter their biological properties.
Mode of Action
Phosphatase removes a phosphate group from its substrate by hydrolysing phosphoric acid monoesters into a phosphate ion and a molecule with a free hydroxyl group. Protein kinases are the effectors of phosphorylation and catalyse the transfer of a y-phosphate from ATP to specific amino acids on proteins. The addition of a phosphate (PO4) molecule to a polar R group of an amino acid residue can turn a hydrophobic portion of a protein into a polar and extremely hydrophilic portion of molecule. In this way it can introduce a conformational change in the structure of the protein via interaction with other hydrophobic and hydrophilic residues in the protein. Several protein kinases are important in cellular control (e.g. glycogen synthase kinase-3, acetyl CoA carboxylase kinase, tyrosine hydroxylase kinase and casein kinase-2), which are themselves controlled by allosteric effectors, phosphorylation, insulin and other growth factors, or by regulators. Protein phosphatase catalytic units are responsible for dephosphorylating many regulated proteins in the cytoplasm that are phosphorylated on serine and threonine residues. Some protein phosphatases are controlled by second messengers. PP-1(Protein phosphatase-1) is regulated by cyclic AMP in several ways that vary with the form of the enzyme and the tissue. It is inhibited by cyclic AMP through the phosphorylation of inhibitor-1 and its isoforms through the phosphorylation of targeting proteins such as the glycogen-binding subunit, and through allosteric inhibition by phosphorylase a. PP-2B (Protein phosphatase-2B) is activated by Ca2+ through the interaction of this second messenger with an integral Ca2+ -binding subunit, as well as calmodulin itself. Protein phosphorylation-dephosphorylation is the basis of a network of interlocking systems that allow hormones and other extracellular signals, acting through just a few second messengers, to coordinate biochemical functions 5.
Functions
Reversible phosphorylation of proteins is an important regulatory mechanism that occurs in living cells 6. Reversible phosphorylation results in a change in conformation the structure in many enzymes and receptors, causing them to become activated or deactivated.
Regulatory roles, the p53 protein is heavily regulated through phosphorylation sites, it has 18 different phosphorylation sites. Activation and phosphorylation of p53 can lead to cell cycle arrest, which can be reversed under some circumstances, or apoptotic cell death 7. In energy-requiring reactions, phosphorylation of Na+/K+-ATPase during the transport of sodium (Na+) and potassium(K+) ions across the cell membrane in osmoregulation to maintain homeostasis.
Enzyme regulation, phosphorylation of the enzyme GSK-3 by AKT (Protein kinase B) is a important regulation in insulin signaling pathway.
Protein-protein interaction, phosphorylation of the cytosolic components of NADPH oxidase, a large membrane-bound, multi-protein enzyme plays an important role in the regulation of protein-protein interactions of the enzyme.
Protein degradation, phosphorylation of some proteins causes them to be degraded by the ATP-dependent ubiquitin/proteasome pathway. These proteins become substrates for particular E3 ubiquitin ligases only when they are phosphorylated 8.
References
1. Levene PA, Alsberg CL (1906). The cleavage products of vitellin. J. Biol. Chem., 2(1): 127-133.
2. Lipmann FA, Levene PA (1932). Serinephosphoric acid obtained on hydrolysis of vitellinic acid. J. Biol. Chem., 98 (1):109-114.
3. Burnett G, Kennedy EP (1954). The enzymatic phosphorylation of proteins. J. Biol. Chem., 211(2):969–980.
4. Aumailley M, Bruckner-Tuderman L, Carter WG, Deutzmann R, Edgar D, Ekblom P, Engel J, Engvall E, Hohenester E, Jones JC, Kleinman HK, Marinkovich MP, Martin GR, Mayer U, Meneguzzi G, Miner JH, Miyazaki K, Patarroyo M, Paulsson M, Quaranta V, Sanes JR, Sasaki T, Sekiguchi K, Sorokin LM, Talts JF, Tryggvason K, Uitto J, Virtanen I, von der Mark K, Wewer UM, Yamada Y, Yurchenco PD (2005). A simplified laminin nomenclature. Matrix Biol., 24(5):326-332.
5. Cohen P (1988). Protein Phosphorylation and Hormone Action. Proceedings of the Royal Society of London. Biological Sciences, 234(1275):115-144.
6. Barford D, Das AK, Egloff MP (1998). The structure and mechanism of protein phosphatases: insights into catalysis and regulation. Annu Rev Biophys Biomol Struct., 27:133–164.
7. Ashcroft M, Kubbutat MH, Vousden KH (1999). Regulation of p53 function and stability by phosphorylation. Mol. Cell. Biol., 19(3):1751–1758.
8. Babior BM (1999). NADPH oxidase: an update. Blood, 93(5):1464–1476.
| DOI | 名称 | |
|---|---|---|
| 10.3390/metabo6040035 | Metabolomic Profiling of the Effects of Melittin on Cisplatin Resistant and Cisplatin Sensitive Ovarian Cancer Cells Using Mass Spectrometry and Biolog Microarray Technology | 下载 |
| 10.1016/0005-2760(93)90292-h | Responses of purified phospholipases A2 to phospholipase A2 activating protein (PLAP) and melittin | 下载 |
| 10_1002cplu_202200240 | Self-Assembly of Linear, Natural Antimicrobial Peptides: An Evolutionary Perspective | 下载 |





